Affinity, also known as Critical Chance, in Monster Hunter World (MHW) is a weapon's chance to deal a bonus or a penalty to Damage on attacks. Some Weapons have innate Affinity, but it can also be increased with Skills and Equipment For a more complete rundown on combat with Monsters. 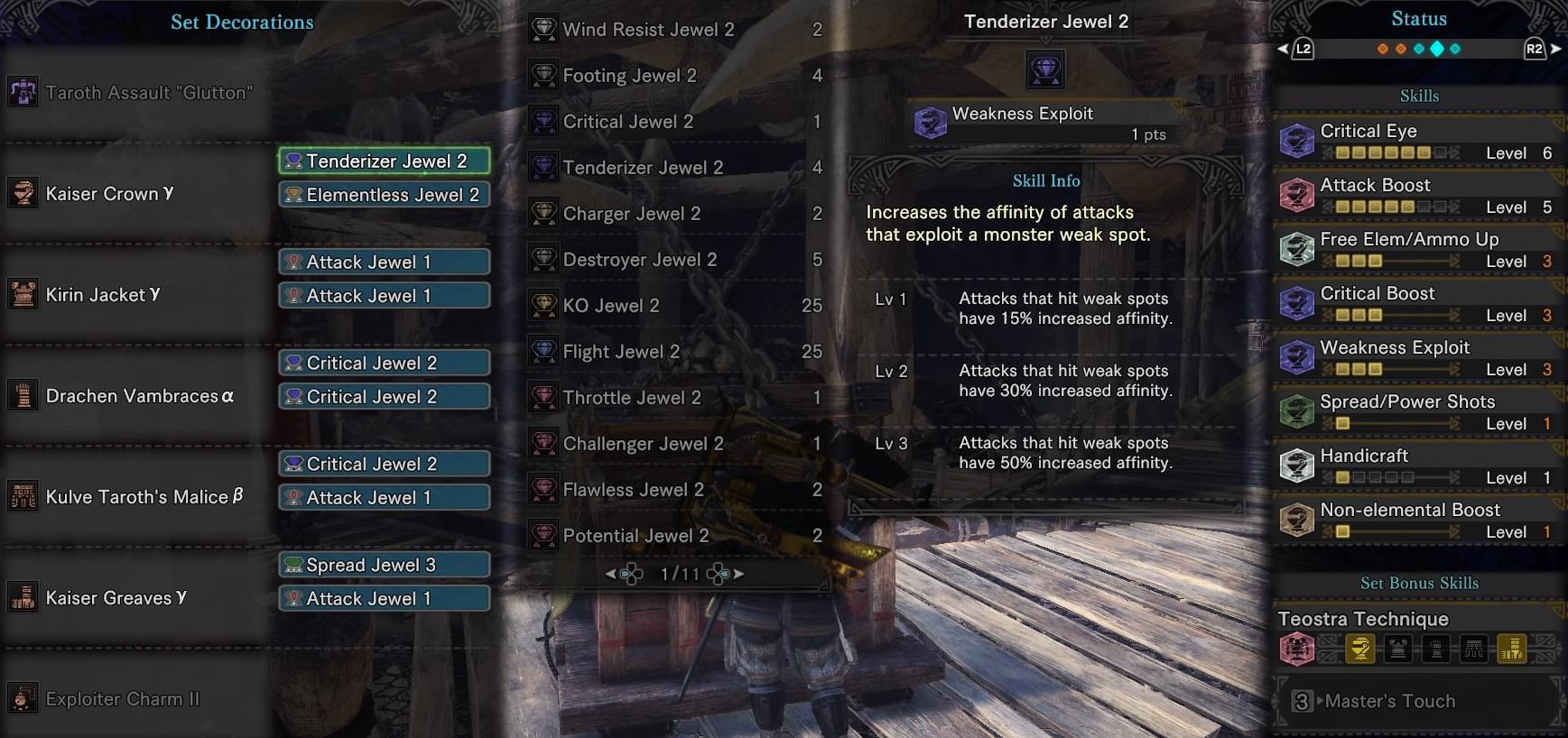 Feb 08, 2018 Increases Affinity for attacks that hit a monster’s weak spots by 15% at level 1, 30% at level 2 and 50% at level 3. Experiment with Affinity and you may find some weapon and armor combinations that turn you into a death-dealing damage machine. Good luck and happy hunting. If you're interested in Monster Hunter World. Feb 15, 2018 While there are tons of different skills in Monster Hunter World, quite a few of them emphasize affinity – boosting the stat directly or in certain conditions. And now that players know what the stat does they can effectively maximize their build for damage. Monster Hunter World is out now for PS4 and Xbox One.
Feb 08, 2018 Increases Affinity for attacks that hit a monster’s weak spots by 15% at level 1, 30% at level 2 and 50% at level 3. Experiment with Affinity and you may find some weapon and armor combinations that turn you into a death-dealing damage machine. Good luck and happy hunting. If you're interested in Monster Hunter World. Feb 15, 2018 While there are tons of different skills in Monster Hunter World, quite a few of them emphasize affinity – boosting the stat directly or in certain conditions. And now that players know what the stat does they can effectively maximize their build for damage. Monster Hunter World is out now for PS4 and Xbox One.

Nickel(II) hydroxide has two well-characterized polymorphs, α and β. The α structure consists of Ni(OH) 2 layers with intercalated anions or water. The β form adopts a hexagonal close-packed structure of Ni 2+ and OH − ions. In the presence of water, the α polymorph typically recrystallizes to the β form. Mar 30, 2009 2.2. Growth of carbon nanotubes. Carbon nanotubes were produced by pyrolyzing Ni(dmg) 2 at temperatures: 750, 800, 850, 900, 950 and 1000 °C, respectively, under Ar atmosphere. In a typical experiment, a sample of Ni(dmg) 2 (0.2 g) was placed into a crucible and introduced into one end of a silica tube (ID: 20 mm, length 112 cm). The tube was.
| Names | |
|---|---|
| IUPAC name | |
Other names
| |
| Identifiers | |
| |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.002.201 |
| EC Number | |
PubChemCID | |
| RTECS number |
|
| UNII | |
CompTox Dashboard(EPA) | |
| |
| |
| Properties | |
| C4H8N2O2 | |
| Molar mass | 116.120 g·mol−1 |
| Appearance | White/Off White Powder |
| Density | 1.37 g/cm3 |
| Melting point | 240 to 241 °C (464 to 466 °F; 513 to 514 K) |
| Boiling point | decomposes |
| low | |
| Structure | |
| 0 | |
| Hazards | |
| Main hazards | Toxic, Skin/Eye Irritant |
| Safety data sheet | External MSDS |
| GHS pictograms | |
| GHS Signal word | Danger |
| H228, H301 | |
| P210, P240, P241, P264, P270, P280, P301+310, P321, P330, P370+378, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
| Hydroxylamine salicylaldoxime | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| verify (what is ?) | |
| Infobox references | |
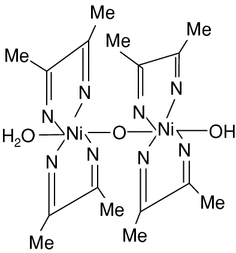
If the pH does become too low the equilibrium of the above reaction favors the formation of the nickel (II) ion, causing the dissolution of Ni (DMG) 2 back into the mother liquor. A slight excess of the reagent has no action on the precipitate, but a large excess should be avoided because of the possible precipitation of the reagent itself. Nickel(II) is square planar. It is surrounded by two equivalents of the conjugate base (dmgH −) of dimethylglyoxime (dmgH 2). The pair of organic ligands are joined through hydrogen bonds to give a macrocyclic ligand. Nickel-dimethylglyoxime complex (abbreviated as Ni(II)(DMG)2) modified carbon paste and graphite electrodes were prepared by mixing Ni(II)(DMG)2 with graphite paste, and coating Ni(II)(DMG)2 to the graphite surface. It is necessary to cycle the electrode potential to a high value (e.g. 0.8V versus SCE) for the preparation of the modified electrodes.
However, Mac doesn’t allow us to install their operating system on Windows that’s why we have the software and through that, we can install any Mac Operating System.The installation is quite simple and before going to install MacOS High Sierra, or on your PC. 

Dimethylglyoxime is a chemical compound described by the formula CH3C(NOH)C(NOH)CH3. Its abbreviation is dmgH2 for neutral form, and dmgH for anionic form, where H stands for hydrogen. This colourless solid is the dioxime derivative of the diketone butane-2,3-dione (also known as diacetyl). DmgH2 is used in the analysis of palladium or nickel. Its coordination complexes are of theoretical interest as models for enzymes and as catalysts. Many related ligands can be prepared from other diketones, e.g. benzil.
Preparation[edit]
Dimethylglyoxime can be prepared from butanone first by reaction with ethyl nitrite to give biacetyl monoxime. The second oxime is installed using sodium hydroxylamine monosulfonate:[1]
Complexes[edit]
Dimethylglyoxime is used to detect and quantify nickel, which forms the bright red complex nickel bis(dimethylglyoximate) (Ni(dmgH)2). The reaction was discovered by L. A. Chugaev in 1905.[2]
Cobalt complexes have also received much attention. In chloro(pyridine)cobaloxime[3] the macrocycle [dmgH]22− mimics the macrocyclic ligand found in vitamin B12.
Structure of chloro(pyridine)cobaloxime.
Ni Dmg 2 2 Structures
References[edit]
Ni(dmg)2 Structure
- ^Semon, W. L.; Damerell, V. R. (1930). 'Dimethylglyoxime'. Organic Syntheses. 10: 22. doi:10.15227/orgsyn.010.0022.CS1 maint: multiple names: authors list (link)
- ^Lev Tschugaeff (1905). 'Über ein neues, empfindliches Reagens auf Nickel'. Berichte der Deutschen Chemischen Gesellschaft. 38 (3): 2520–2522. doi:10.1002/cber.19050380317.
- ^Girolami, G. S.; Rauchfuss, T.B.; Angelici, R. J. (1999). Synthesis and Technique in Inorganic Chemistry: A Laboratory Manual (3rd ed.). pp. 213–215.
Ni Dmg 2 2 Structure Diagram
Retrieved from 'https://en.wikipedia.org/w/index.php?title=Dimethylglyoxime&oldid=931825982'
